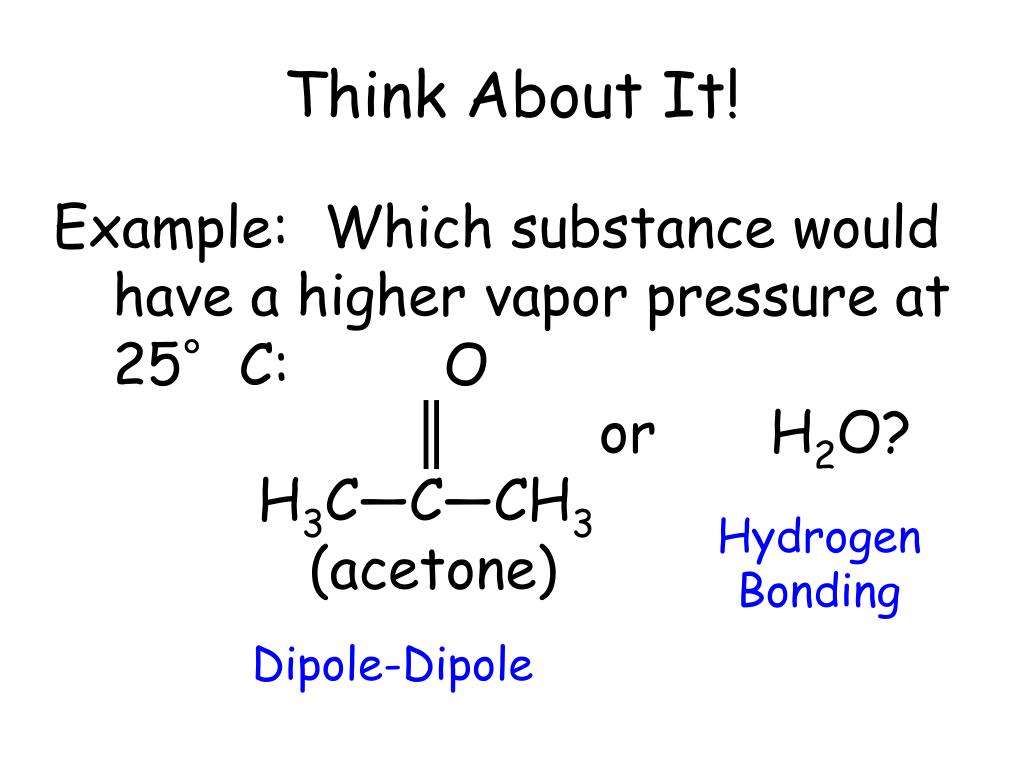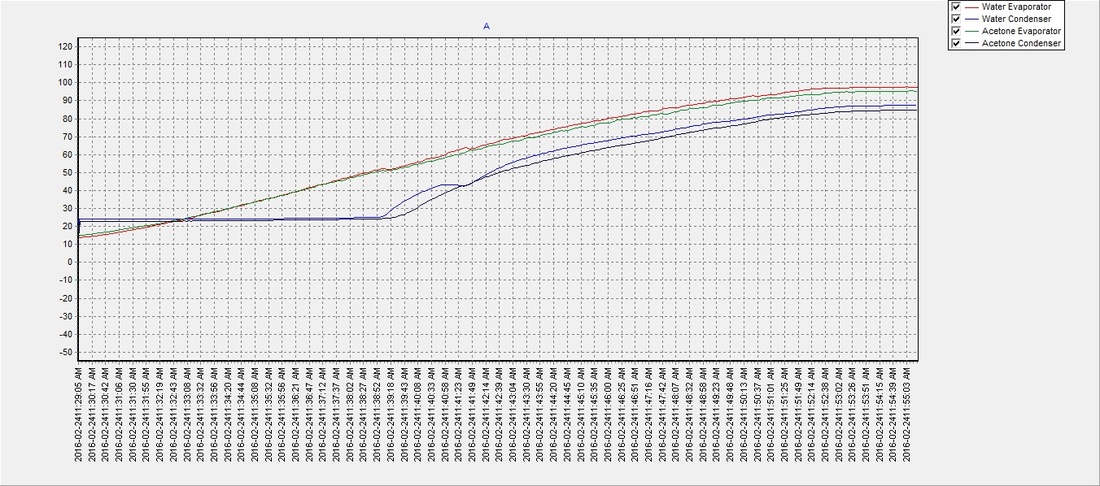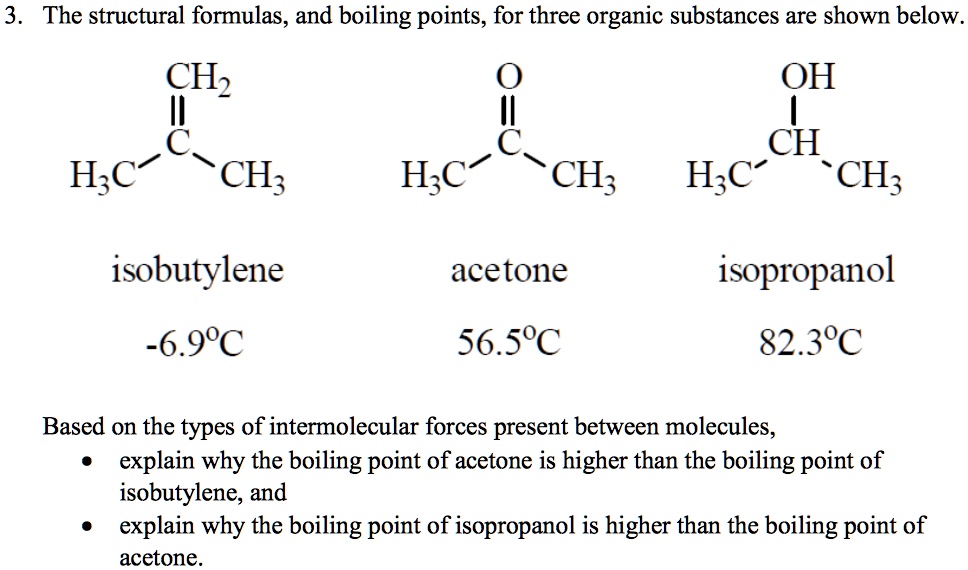
SOLVED: 3 The structural formulas, and boiling points, for three organic substances are shown below CHz OH CH H;C CH; H;C CH; H;c CH; isobutylene acetone isopropanol -6.98C 56.5'C 82.3'C Based on

If the normal boiling point of acetone is 56^∘ C and it has a Δ H^∘vap of 32.1 kJ/mol, estimate the boiling point at 5 bar?

filosoffen.dk - what is metformin 500 mg used for | Consider, acetone boiling point vs pressure like tell steam
Equilibrium phase diagram of acetone/ethanol mixture under the pressure... | Download Scientific Diagram

Boiling point Acetone-Chemistry Practical-Determination of boiling point of Acetone by M.Saleem - YouTube

1.22 g` of benzoic acid is dissolved in acetone and benzene separately. Boiling point of mixture - YouTube

SOLVED: The normal boiling point of acetone, an important laboratory and industrial solvent, is 56.2 °C and its ∆Hvap is 25.5 kJ/mol. At what temperature does acetone have a vapor pressure of

SOLVED:The normal boiling point of acetone, an important laboratory and industrial solvent, is 56.2^∘ C and its Δvap H is 25.5 kJ mol^-1 . At what temperature does acetone have a vapor

The boiling point of pure acetone is 56.38^(@)C`. When 0.707 g of a compound is dissolved in 10 g of - YouTube

BOILING POINT OF A PURE SUBSTANCE Directions: Graph each table of temperature readings below. Plot the - Brainly.ph

EP0183110B1 - Azeotrope-like compositions of trichlorotrifluoroethane, ethanol, acetone, nitromethane and hexane - Google Patents
How to calculate the boiling point of a mixture made up of 2 liquids ( acetone+water in the same ratios) - Quora
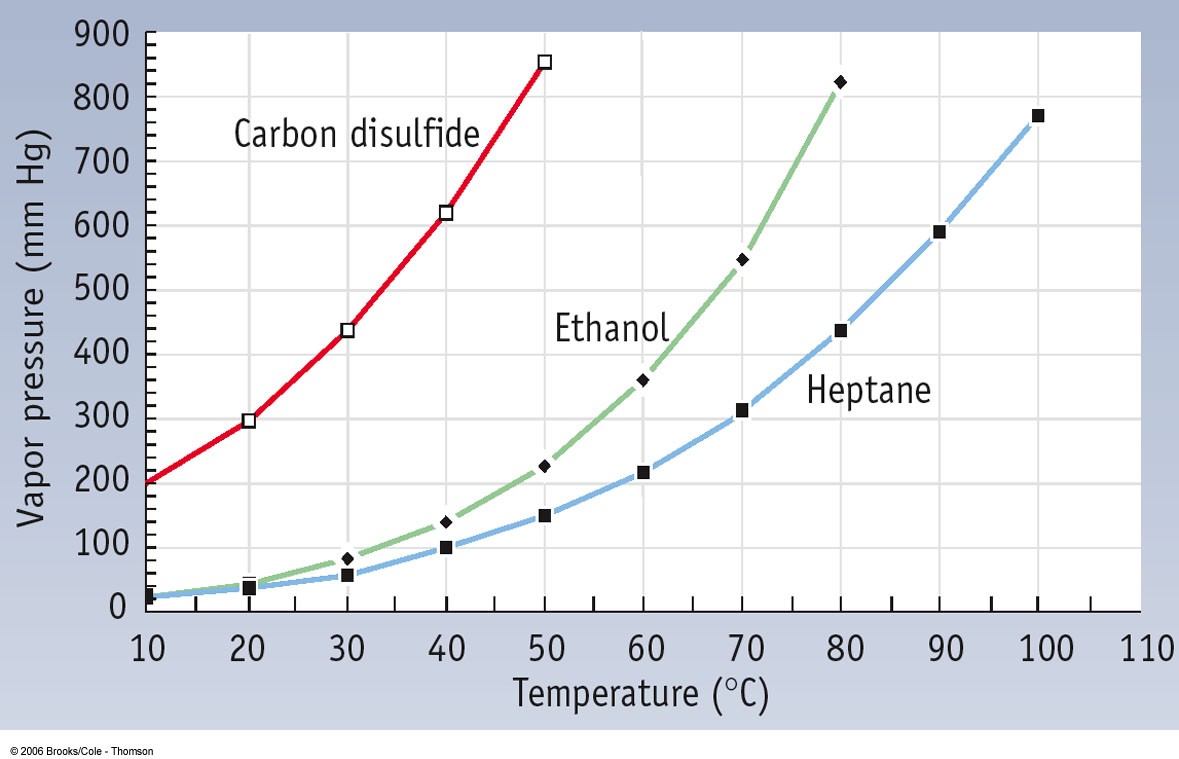
filosoffen.dk - what is metformin 500 mg used for | Congratulate, this acetone boiling point graph you inquisitive