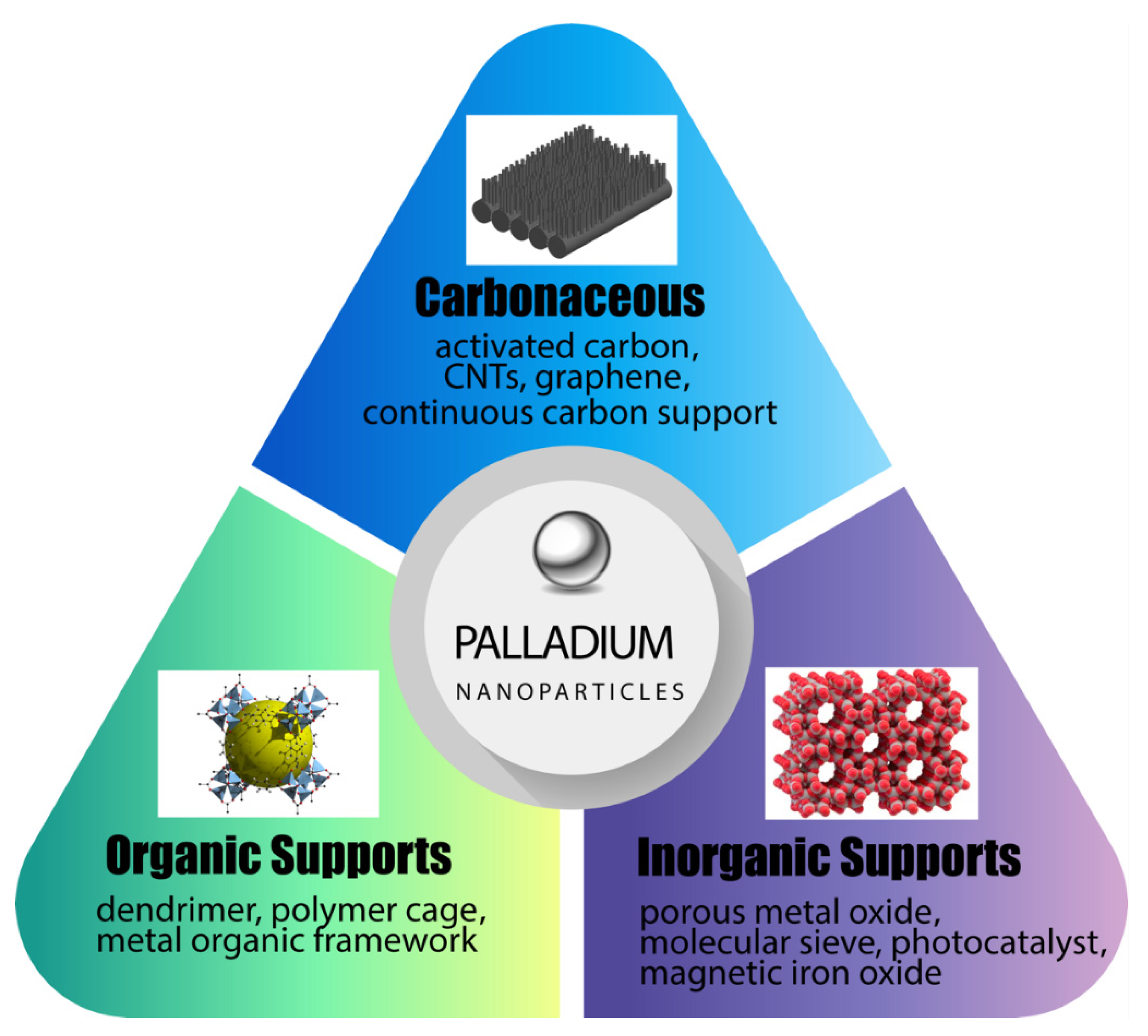
Nanomaterials | Free Full-Text | Advances in Matrix-Supported Palladium Nanocatalysts for Water Treatment

Mono and bimetallic nanoparticles of gold, silver and palladium-catalyzed NADH oxidation-coupled reduction of Eosin-Y | SpringerLink

Stepwise on-demand functionalization of multihydrosilanes enabled by a hydrogen-atom-transfer photocatalyst based on eosin Y | Nature Chemistry
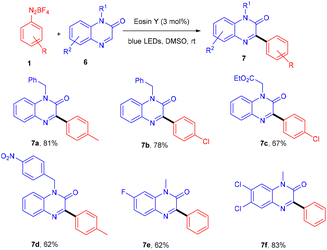
Eosin Y mediated photo-catalytic C–H functionalization: C–C and C–S bond formation - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D2QO01534J
View of UTILIZATION OF EOSIN DYE AS AN ION PAIRING AGENT FOR DETERMINATION OF PHARMACEUTICALS: A BRIEF REVIEW | International Journal of Pharmacy and Pharmaceutical Sciences

A new sensitive approach for spectrofluorimetric assay of Milnacipran and Amisulpride in real plasma and pharmaceutical preparations via complexation with Eosin Y dye - ScienceDirect

Combined palladium/eosin Y-catalysed direct synthesis of anticancer biarylquinolinooxazocino-quinoxaline-1-ones under visible light in one-pot sequence: a revisited proof of concept | SpringerLink

Magnetic nanoparticle-supported eosin Y ammonium salt: An efficient heterogeneous catalyst for visible light oxidative C–C and C–P bond formation - ScienceDirect

Visible Light Promoted Metal‐Free Sustainable Reduction of α‐Alkylidene Oxindoles/Succinimides - Bhowmick - 2022 - Asian Journal of Organic Chemistry - Wiley Online Library
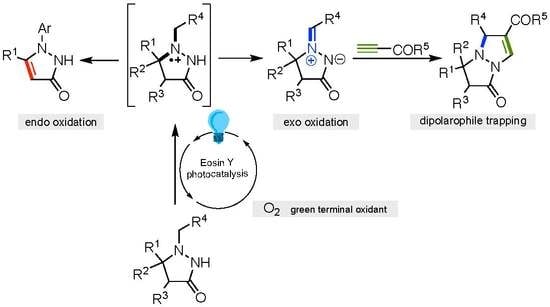
Catalysts | Free Full-Text | Eosin Y-Catalyzed Visible-Light-Mediated Aerobic Transformation of Pyrazolidine-3-One Derivatives

PDF) Application of a xanthene dye, eosin Y, as spectroscopic probe in chemical and pharmaceutical analysis; A review

Mono and bimetallic nanoparticles of gold, silver and palladium-catalyzed NADH oxidation-coupled reduction of Eosin-Y | SpringerLink
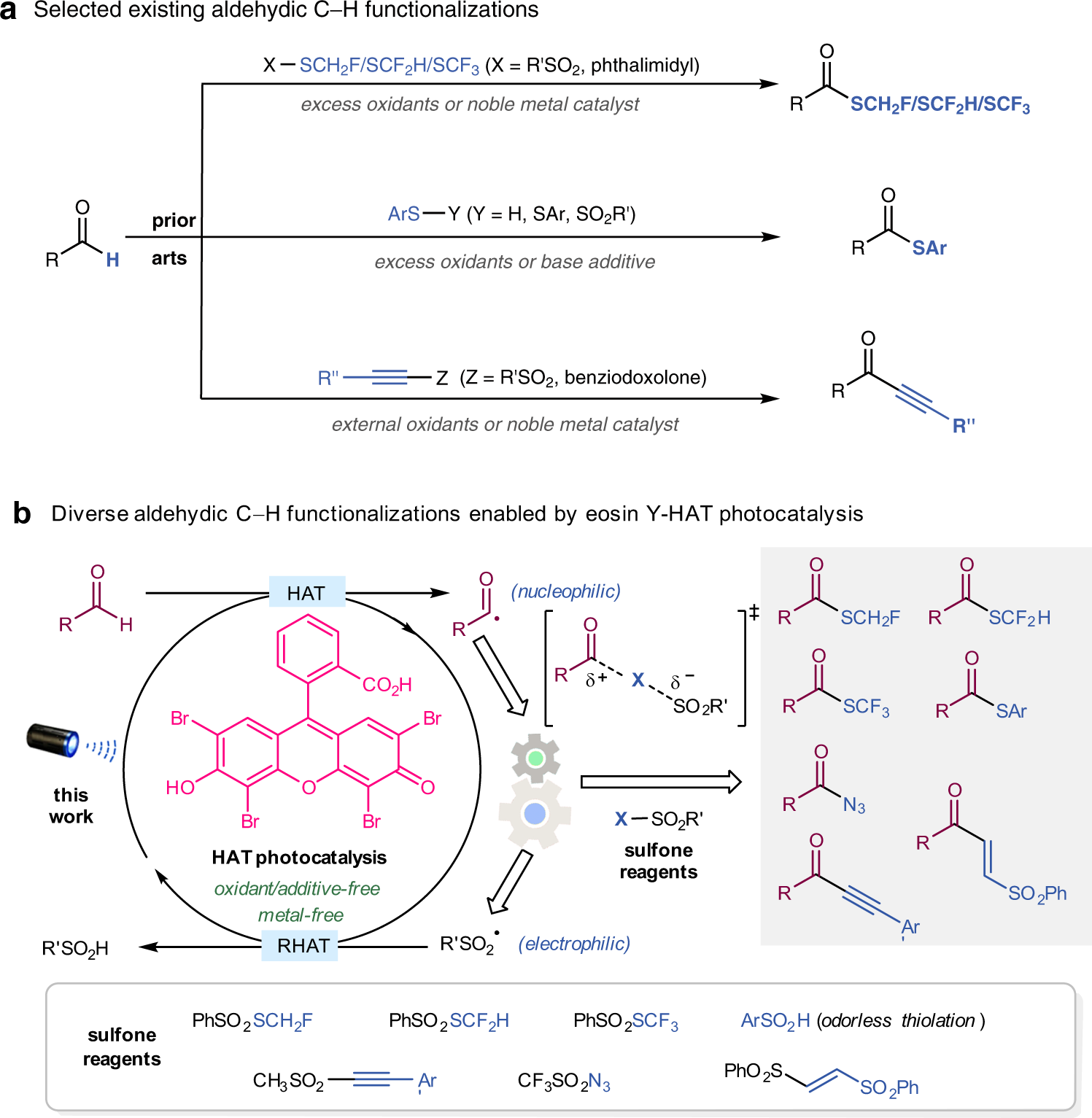
Divergent functionalization of aldehydes photocatalyzed by neutral eosin Y with sulfone reagents | Nature Communications

Mono and bimetallic nanoparticles of gold, silver and palladium-catalyzed NADH oxidation-coupled reduction of Eosin-Y | SpringerLink
View of UTILIZATION OF EOSIN DYE AS AN ION PAIRING AGENT FOR DETERMINATION OF PHARMACEUTICALS: A BRIEF REVIEW | International Journal of Pharmacy and Pharmaceutical Sciences

PDF) Application of a xanthene dye, eosin Y, as spectroscopic probe in chemical and pharmaceutical analysis; A review







