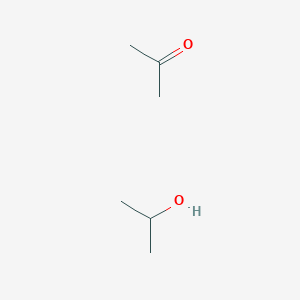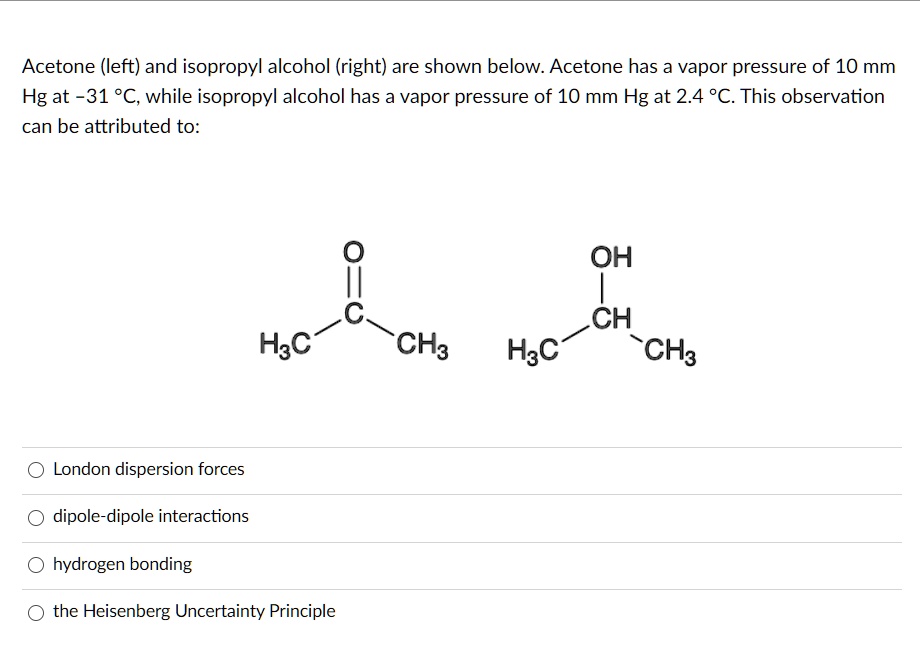
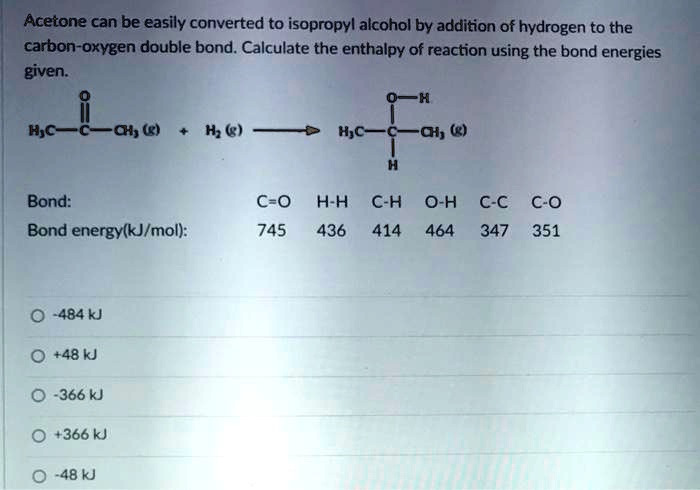
SOLVED: Acetone (left) and isopropyl alcohol (right) are shown below: Acetone has a vapor pressure of 10 mm Hg at -31 °C, while isopropyl alcohol has a vapor pressure of 10 mm
a) The equilibrium of a redox reaction between isopropyl alcohol and... | Download Scientific Diagram

Oxidation Of Isopropyl Alcohol, Acetone, And Butyl Compound By Nuetral And Alkaline Potasium ......: Sefton, Lily Bell: 9781271780822: Amazon.com: Books

Convert: (i) 3 - phenyl - 1 - propanol to 3 - phenyl - 1 - propanal.(ii) isopropyl alcohol to acetone.
Vector set of most common organic solvents with structural formula. Dichloromethane, acetone, benzene, ethanol, toluene, methanol, isopropyl alcohol, water, acetic acid. Molecules isolated on white. Stock Vector | Adobe Stock
Amaranth's Castaways: Face Up Removal: Windsor and Newton vs Isopropyl Alcohol vs Nail Polish Remover
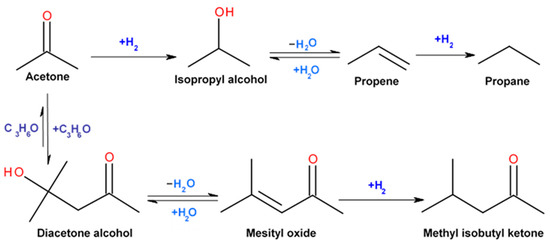
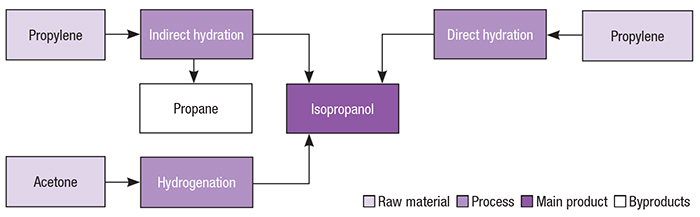
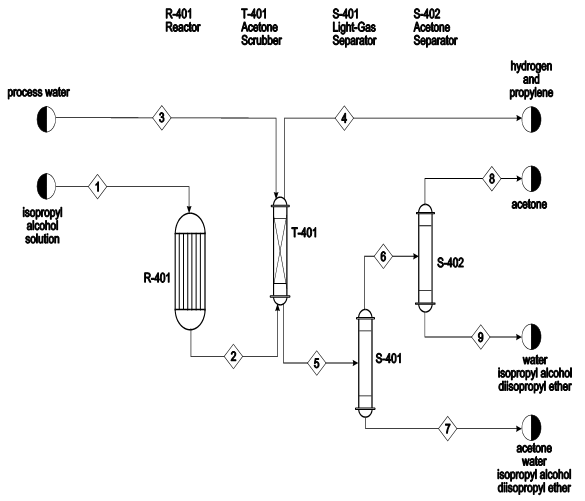
Catalysts | Free Full-Text | Highly Selective Gas-Phase Catalytic Hydrogenation of Acetone to Isopropyl Alcohol
Electrochemical Hydrogenation of Acetone to Produce Isopropanol Using a Polymer Electrolyte Membrane Reactor

Evaporation monitoring curves of Isopropyl Alcohol, Acetone and Ethanol. | Download Scientific Diagram
![PDF] Detection of isopropyl alcohol in a patient with diabetic ketoacidosis. | Semantic Scholar PDF] Detection of isopropyl alcohol in a patient with diabetic ketoacidosis. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/39b6f0e499270b9261c865ee3904fa36a919144e/3-Figure1-1.png)