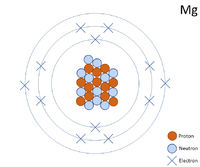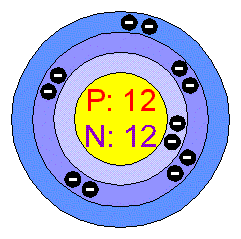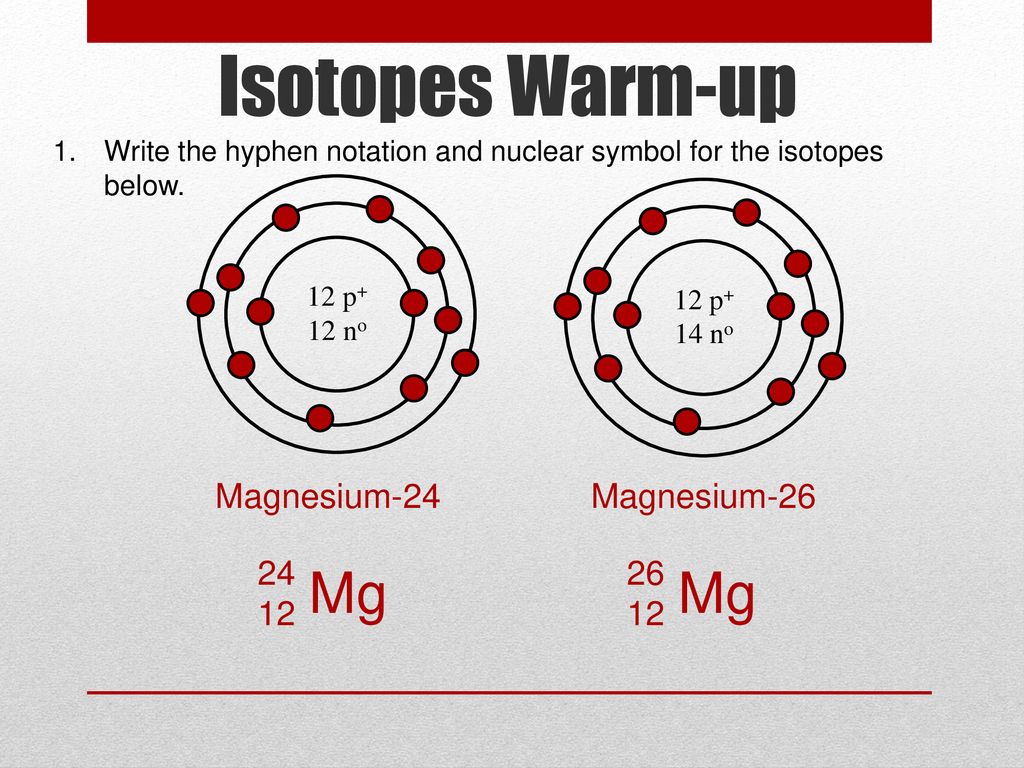
Isotopes Warm-up Write the hyphen notation and nuclear symbol for the isotopes below. 12 p+ 12 no 12 p+ 14 no 2. A atom has 26 protons and 30 neutrons. - ppt download

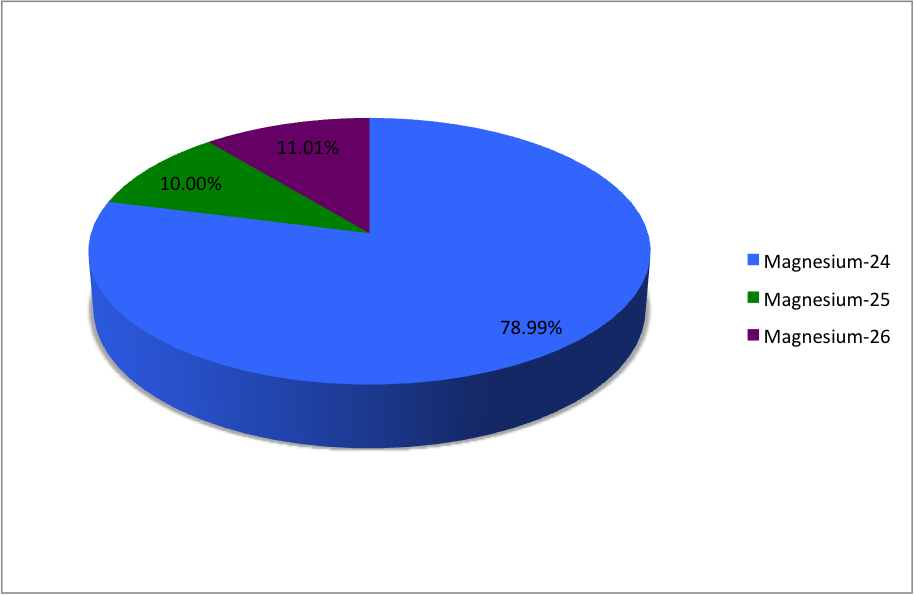
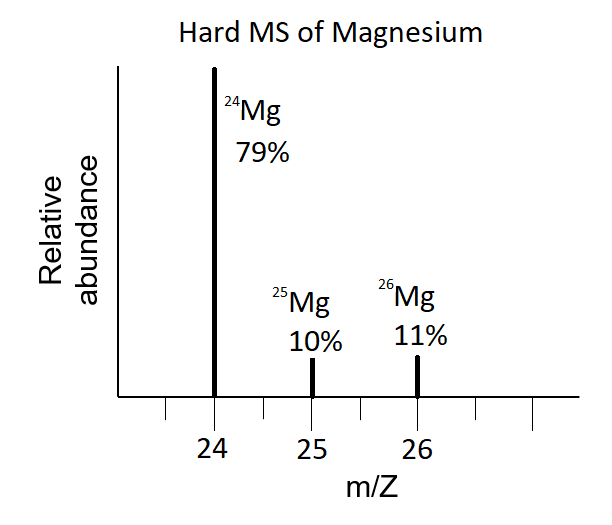
In a periodic table the average atomic mass of magnesium is given as 24.312 u. The average value is based on their relative natural abundance on earth. The three isotopes and their

Average atomic mass of magnesium is `24.31`amu. This magnesium is composed of 79 mole % of `24mg... - YouTube

Amazon.com: GoodSense Esomeprazole Magnesium Delayed Release Capsules 20 mg, Acid Reducer, Treats Heartburn, 28 Count : Health & Household