What is the Vant Hoff's factor, and can you give the derivation of the formula for association and dissociation in solutions? - Quora

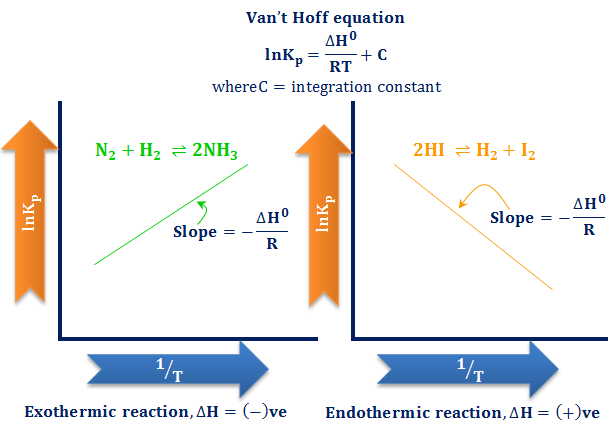
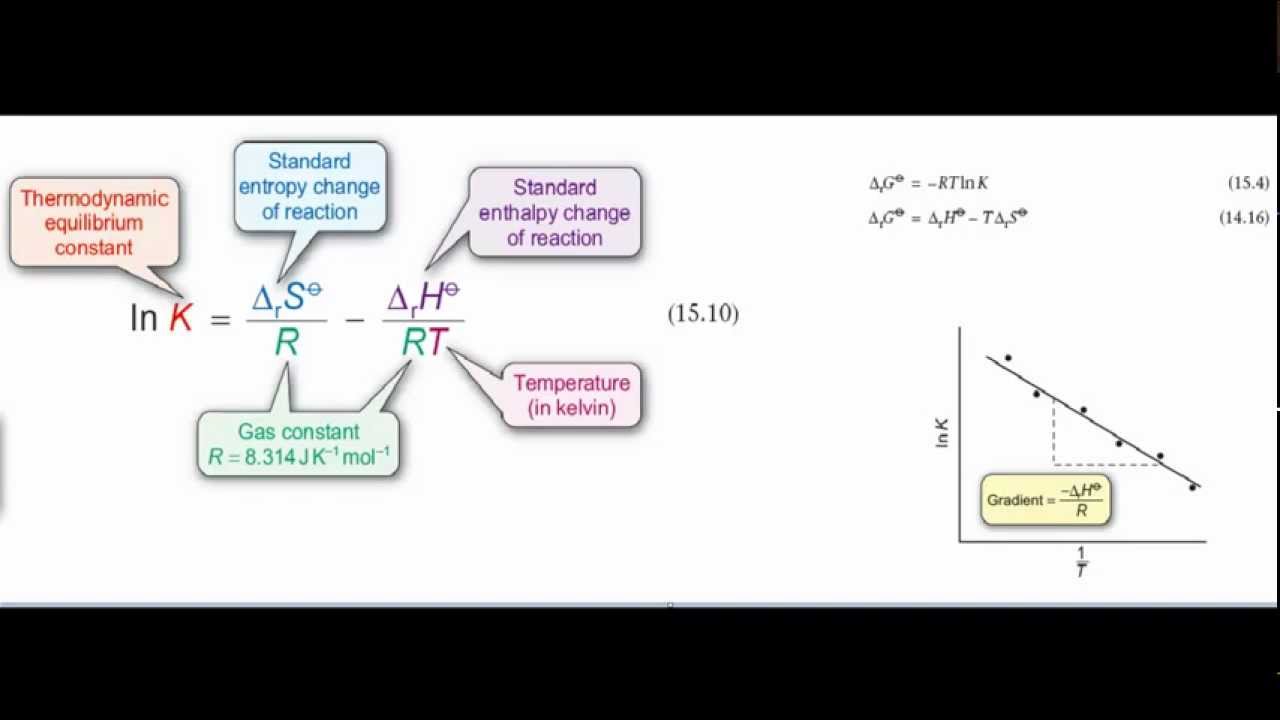
van't Hoff's equation gives the quantitative relation between change in value of K with change in temperature.

PDF) Van't Hoff equation-equilibrium How much Van't Hoff equation -effect on temperature | Kajal Panda - Academia.edu